
It's bent with a bond angle of 120 degrees. And that's it, the SO2 molecular geometry. We can remember that this is a bent molecule, or we can look it up on a table. Here we can see AX2N is Bent and has a bond angle of 120 degrees. Well ignore the ones, and we can see that we have AX2N. 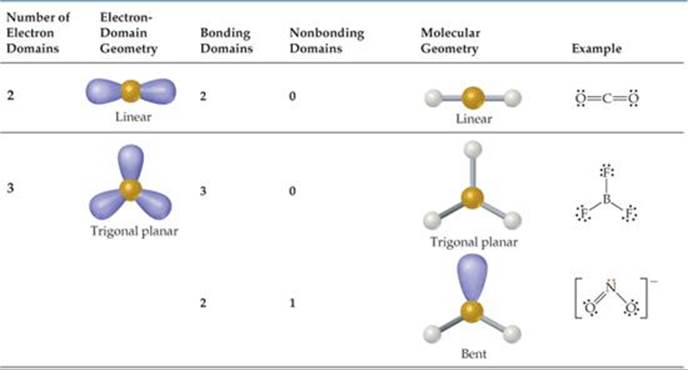
We'll ignore the ones, and we can see that we have AX2N. We can quickly learn the molecular geometry of any substance. The molecular geometry of SO2 is curved, with a bond angle of 120. It causes a repulsion of electron pairs to create the 120-degree angle. We only have one unbonded electron pair for SO2. One single atom of SulphurSulphur is bonded with two atoms of Oxygen covalently. Alternatively, the electron geometry of sulphur dioxide is in the shape of a trigonal. A is the central atom, the Sulfur, we have one of those, X the bonded atoms, those two oxygen atoms, and N that is the unbonded electron pairs. The molecular geometry of SO2 is curved, with a bond angle of 120. We can also use the AXN notation to determine the shape of SO2. 
In essence the Valence Shell Electron Pairs are Repelling each other, giving SO2 it's shape. The Lewis structure doesn't tell us this directly but if we visualize the atoms and unbonded electron pairs on that central atom, repelling each other, we'll see that oxygen atoms move away from the unbonded pair and give the molecule a bent shape. The bond angle is proportional to the A / B ratio of atomic polarizabilities ( Table 1 b, Table 2 b). , SO2 Lewis structure, Molecular geometry, Bond angle, Shape., What is the. This is because electrons are negative and they repel each other. , SO2 Molecular Geometry,Shape and Bond Angles (Sulfur Dioxide)., SF6. A quick explanation of the molecular geometry of SO3 2- (Sulfite ion) including a description of the SO3 2- bond angles.Looking at the SO3 2- Lewis structure. First, we can use the Lewis structure for SO2 to visualize how the atoms and unbonded electron pairs will spread out in three dimensions. In sulfur dioxide, there are three electron clouds around the sulfur. But it is related to dipole moment and assertion is the explanation from bent angle so here reason is unable to give explanation of assertion.We can find the molecular geometry for SO2 it two ways. The whole molecule is planar, and its shape resembles two triangles joined point to point. S atom in both S O 2 and S O 3 is sp2 -hybridized. The extra repulsion of the lp vs double bonds accounts for reduction to 119. In the case of SO2, there will be a double bond between the sulfur atom (S) and each oxygen atom (O). In S O 2, we have 2 double bonds and one lone pair 3 electron regions so the default angle is 120 degrees. The S O 2 molecule has a dipole moment, The starting point of 109.5 degrees is only applicable for 4 electron regions round the central atom. The bonding in S O 3 can be described as a combination of 3 resonance forms of 1 double and 2 single bonds. So the shape is based on a tetrahedral structure - but the extra repulsion of the non-bonding pair will decrease the O-S-O angles by a few degrees from the 'perfect' tetrahedral angle. It is a nonpolar molecule with a bond angle of 109.5°. Watch this video to know the molecular geometry of Sulfur Dioxide. The molecule comprises one Sulfur Atom and two Oxygen atoms. 
The CH4 Lewis structure comprises two different atoms: carbon and hydrogen. SO2 Molecular Geometry,Shape and Bond Angles (Sulfur Dioxide) Today in this video we are going to share a step-by-step procedure to determine the molecular geometry of SO2 molecules. Methane ( CH4) is a colorless, odorless, and highly combustible gas that is utilized to generate energy and heat houses all over the world. There are 3 bonding sets of electrons and one non-bonding pair on the sulphur. CH4 Lewis Structure & Molecular Geometry. The correct option is B If both assertion and reason are CORRECT, but reason is NOT THE CORRECT explanation of the assertion.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
